|
Facilities |
|
Molecular Photodynamics and Electronics Photonics&Materials Group |
|
MOLECULAR MACHINES
The ability to construct and manipulate molecular systems on the atomic level is the key to creating molecular machines and switches.
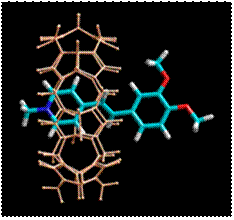
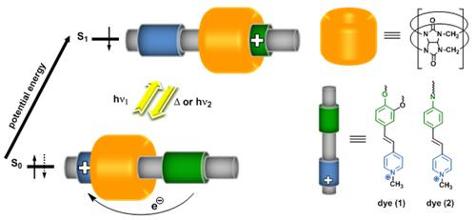
In our studies, we show that simple cationic styryl compounds form highly stable inclusion complexes with a macrocyclic cucurbit[7]uril (CB[7]) host, that can be switched by using light between two states with different shuttling properties. The photoswitchable system is simple and novel compared to the photoswitchable ternary complex between CB[7], Ru(bpy)3 and viologen derivatives reported elsewhere. Switching is anticipated to rely on the photoinduced Charge Transfer (CT) process. Our expectation was that the main binding site in styryl dye for CB[7] is positive charged pyridinium residue while the photoinduced CT will shift the CB[7] to nitrogen or oxygen atom in aryl nucleus whose become the positively charged in photoinduced exited state. The remove of light excitation returns molecules to charge distribution of ground state. This also returns the CB[7] molecule to the position on heterocyclic residue.
The formation and behavior of the inclusion complex of the photoactive molecules with cucurbit[n]urils provides basic understanding for the future design of light-driven elements for advanced molecular machines and optical molecular devices or nanoflappers in systems for example for drug delivery and release.
Copyright 2020 © Gediminas Jonusauskas, Laboratoire Ondes et Matière d’Aquitaine - UMR 5798 CNRS/Université Bordeaux |