I am Maître de Conférences in the Laboratoire Ondes et Matière d’Aquitaine (LOMA) at Université de Bordeaux., France. I am a member of the Soft Matter and Biophysics Team, Mecanogenetics of Cells and Tissues Group.
My research at the Physics/Biology interface focuses on understanding how photosynthesis adapts to the environment (mechanical and/or light). I also teach physics courses from Bachelor to Master.
Research
Research
My main line of research at the Physics/Biology interface focuses on understanding how microscopic organisms adapt their properties under environment constrained (mechanical and/or light).

On-going work
Ahmad Badr got a PhD fellowship to initiate the study of photoresponses in confined micro-alga. Contact us to know more about it.
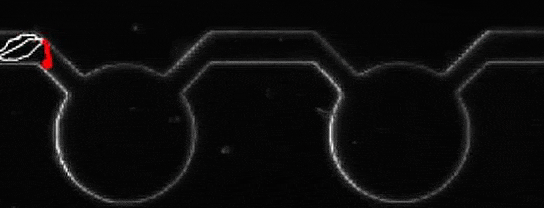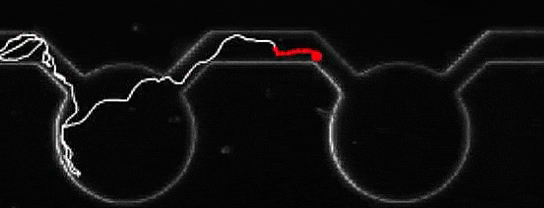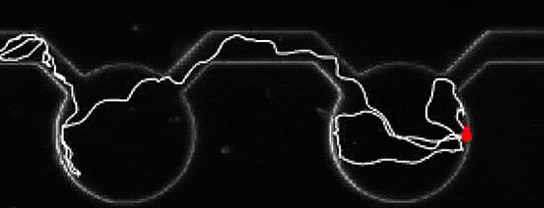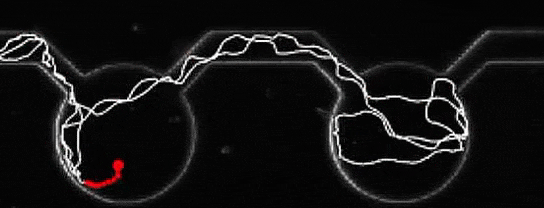
A microalga journey, navigating from pool to pool. Credit: Marco Polin
Publications
Last publications
Role of Hydrodynamics in the Synchronization of Chlamydomonas Flagella
While hydrodynamic coupling has long been considered essential for synchronization of eukaryotic flagella, recent experiments on the unicel
lular biflagellate model organism Chlamydomonas demonstrate that -at the single cell level- intracellular mechanical

coupling is necessary for coordination. It
is therefore unclear what role, if any, hydrodynamic forces actually play in the synchronization of multiple flagella within individual cells, arguably the building block of large scale coordination. Here we address this quest
ion experimentally by transiently blocking hydrodynamic coupling between the two flagella of single Chlamydomonas. Our results reveal that in wild type cells intracellularly mediated forces are necessary and sufficient for flagellar synchronization, with hydrodynamic coupling causing minimal changes in flagellar dynamics. However, fluid-mediated ciliary coupling is responsible for the extended periods of anti-phase synchronization observed in a mutant with weaker intracellular coupling. At the single-cell level, therefore, flagellar coordination depends on a subtle balance between intracellular and extracellular forces.
Authors: Antoine Allard, Krish Desai, Marco Polin and Luc Zorilla.
Enhanced dispersion of active microswimmers in confined flows
This study introduces a unified approach combining microfluidics, Langevin simulations, and statistical analysis to examine how Chlamydomonas reinhardtii, an active swimming microalgae, disperses in confined flows. The findings extend classical Taylor–Aris dispersion theory—originally for passive Brownian particles—

to active swimmers, revealing a transition from ballistic to diffusive behaviors influenced by the flow. Shear flows are shown to impact the swimmers’ dynamics nonlinearly, enhancing spreading in complex ways. These results provide critical insight into microswimmers’ behavior in fluid environments, relevant to both artificial and natural settings. The
work has broader implications for microbial ecology, suggesting ways to understand processes like biofilm formation, nutrient transport, and pathogen dispersal in ecosystems where fluid dynamics shape microorganism movement and interactions.
Authors: Antoine Allard, Yacine Amarouchene, Ahmad Badr, Guirec de Tournemire, Juliette Lacherez, Marc Lagoin and Thomas Salez.
CellMAP: an open source software for Cell Mapping

The Atomic Force Microscopy (AFM) imaging modes relying on force curves yield nanometer-scaled maps of living cells’ morphology and viscoelastic properties. Although AFM manufacturers offer software tools for analyzing cells individually, there is a growing need for fast and accessible tools to compile data from multiple cells into a single dataset, which is lacking within the numerous open-source tools currently available. To address this, we present CellMAP, a user-friendly software tool that streamlines the batch-processing of AFM-derived topography and elasticity maps of living cells, specifically those generated by a JPK-Bruker Nanowizard AFM. Our analysis pipeline starts with data treatment, such as leveling and filtering. Then, it allows measurement of the cell surface, volume, and elasticity distributions, mechanical and geometrical parameters crucial for characterizing cell behavior. CellMAP can also generate a composite cell from a set of standardized cells (e.g., cells constrained by micro-patterns) that reflects the entire population’s behavior.
Authors: Antoine Allard, Clément Campillo, Raphaël Crépin, Guillaume Lamour, Maxime Liboz, Olek Maciejak, Michel Malo and Sid Labdi.
Microbial narrow-escape

Assessing cancer cell characteristics

Authors: Antoine Allard, Clément Campillo, Sid Labdi, Guillaume Lamour, Gaelle Letort, Maxime Liboz, Michel Malo, Juan Pelta and Bénédicte Thiébot.
Members
Members
Current
- Ahmad Badr (2024-2027)
Undergrade
- Alexandre Bernardie (Université de Bordeaux, L3PC)
- Nathan Delage (Université de Bordeaux, L3PC)
- Mikael Gallego (Université de Bordeaux, M2 LMN)
- Shaila Kolomatskaia (Lehigh University, USA)
- Sarah Marquestaut (Université de Bordeaux, BUT Mesures Physiques 3ème année)
Alumni
- Ahmad Badr (Université Paris-Sorbonne, M2 SBCP)
- Elorri Garcia (Université de Bordeaux, M1)
Undergrade
- Louis Linassi (Université de Bordeaux, L3PC 24-25)
- Amal Moumou (Université de Bordeaux, L3PC 24-25)
- Tania Nzong (Université de Bordeaux, L3PC 24-25)
- Mathew Marlowe (Lehigh University, USA)
- Augustin Rebel (Université de Bordeaux, L2PC 23-24)
- Maena Franzinetti (Université de Bordeaux, L3P 23-24)
- Louis Cazenave (Université de Bordeaux, L3PC 23-24)
- Juliette Chapuis (Université de Bordeaux, L3P 22-23)
- Benjamin Fournie (Université de Bordeaux, L3PC 22-23)
- Alex Garcia (Université Paris-Saclay, L3 22-23)
- Pablo Hernandez (Université Paris-Saclay, L3 22-23)
Activities
Activities
Responsabilities
- Since January 2026, L1PC co-responsable
- Organizer of Chlamyting, the Chlamydomonas Biophysical Meeting, May 17th 2024, Bordeaux
- Organiser of BioPhy Journal Club
- Since September 2023: Communication – Physics Department
- Since March 2023: Equality-Parity referent for CNRS and referent for societal and environmental transitions for University of Bordeaux
Curriculum vitae
Curriculum vitae
Here: Curriculum Vitae (sept. 2023).
I obtained a PhD in Physics from Université Paris-Saclay, studying on the role of actin cytoskeleton on lipid membrane nanotubes. This work was developed in collaboration with Clément Campillo in Laboratoire Analyse, Modélisation, Matériaux pour la Biologie et l’Environnement (LAMBE), in Évry and with Cécile Sykes in Laboratoire Physico-Chimie Curie (PCC), Curie Institute, in Paris. This work is freely available online.
For this work, I have been awarded two PhD prices:
- C’Nano 2020 in the Interdisciplinary category (here)
- Groupe d’Études des Membranes (GEM 2021)
I then joined Marco Polin‘s group at the Physics Department at University of Warwick, in England, where I worked on understanding how the model swimming organism Chlamydomonas reinhardtii, a biflagellated microalgae, responds to light stimulus.

Antoine ALLARD
Laboratoire Ondes et Matière d’Aquitaine (LOMA) 351 cours de la libération 33405 Talence Cedex
Office: A4N/003
Phone : + 33 (0)5 40 00 25 41
E-mail: antoine.allard [at] u-bordeaux.fr










